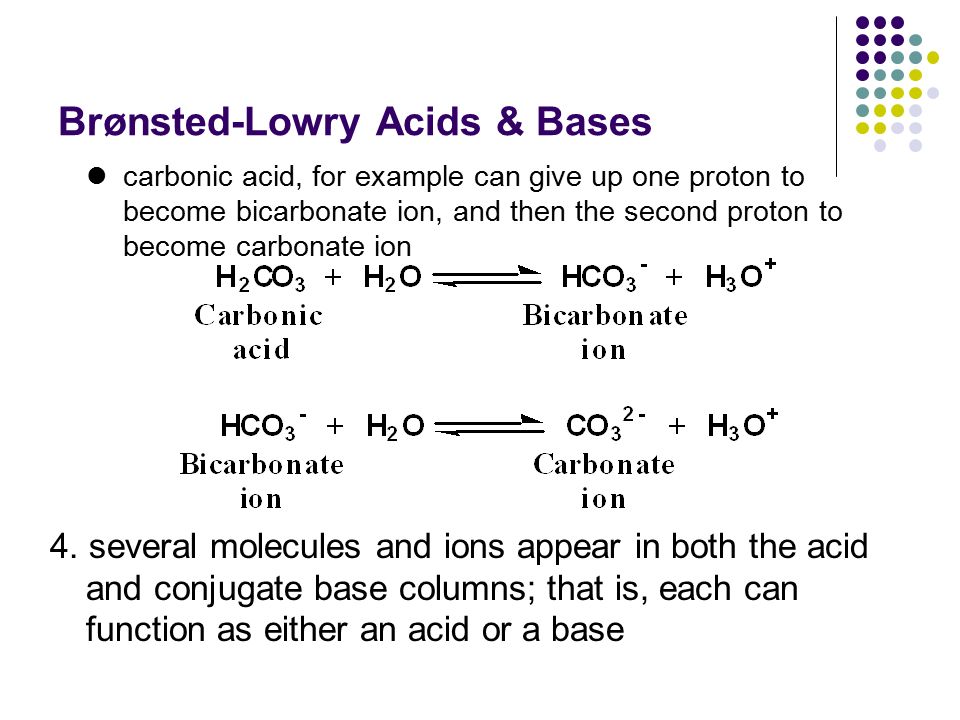
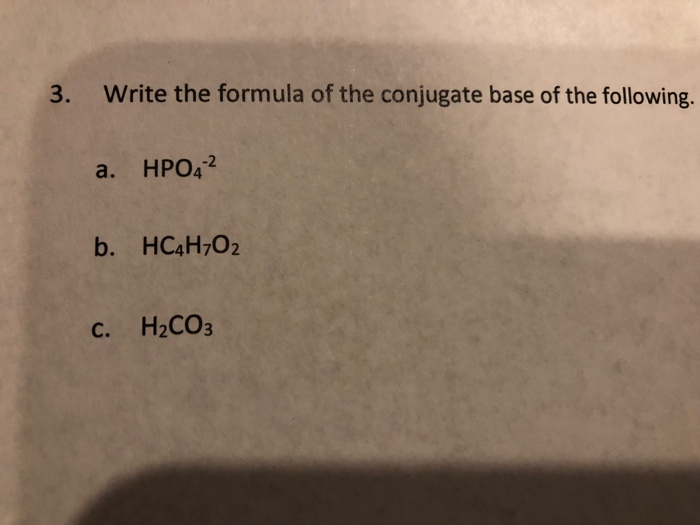SOLVED: QUESTION 11 In the following reaction: HCO3" (aq) + H2O (aq) -> H2CO3 (aq) + OH" (aq) 0 HCO3" is an acid and is H2CO3 its conjugate base. 0 H20 is

a) Mention conjugate base of each of the following: HS^-,H3O^+,H2PO4^-,HSO4^-,HF,CH3COOH,C6H5OH,HClO4,NH4^+ (b) Mention the conjugate acid of each of the following: OH^-,CH3COO^-,Cl^-,CO3^2 - ,H2PO4^-,CH3NH2,CH3COOH,NH2^- (c) Which of the following ...

SOLVED: Konsider the reaction: H2SO3 HCO3" H2CO3 HSO3" a) Identify the acid, base, conjugate acid and conjugate base b. What is the conjugate base of water? C What is the value of [