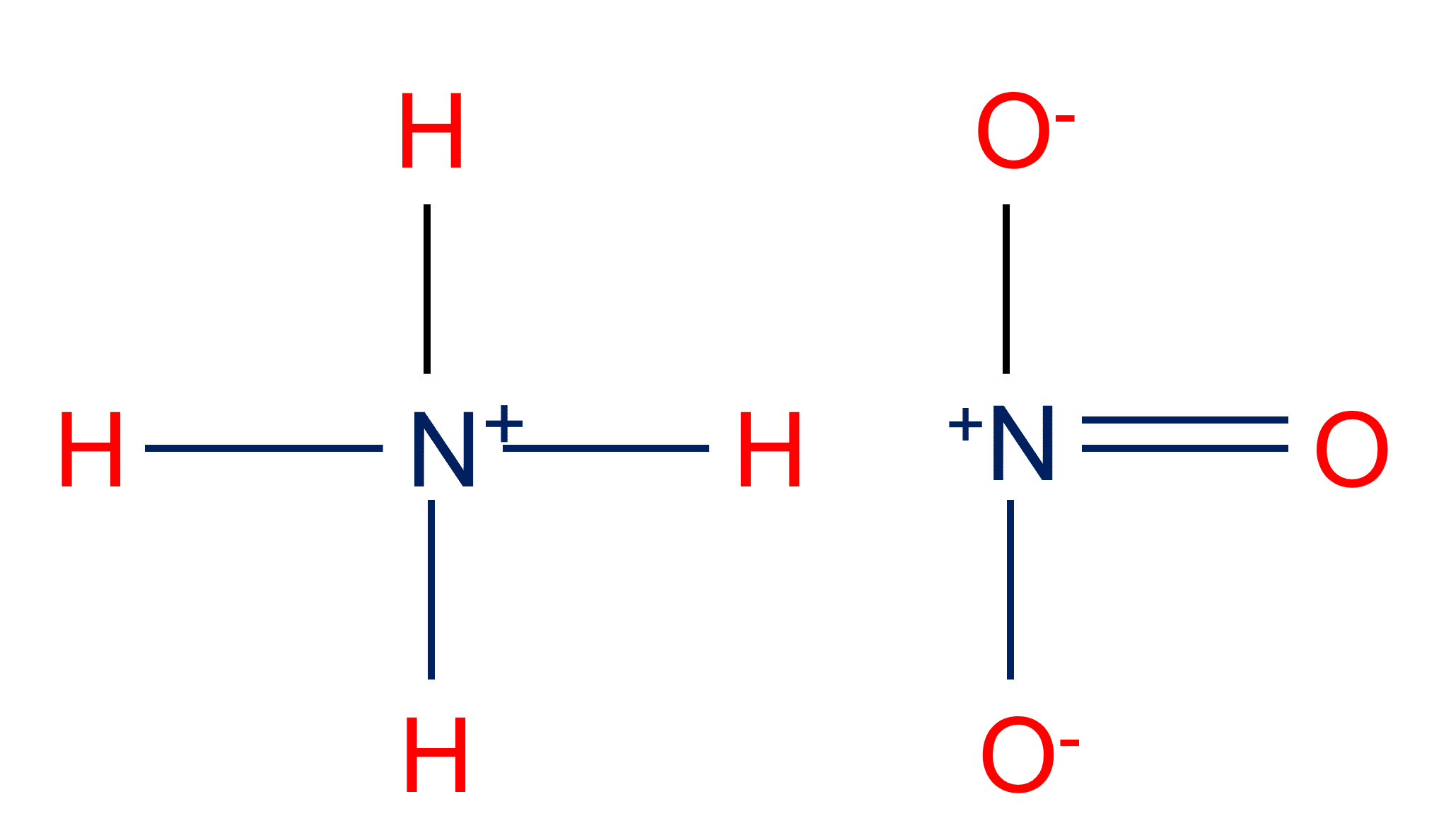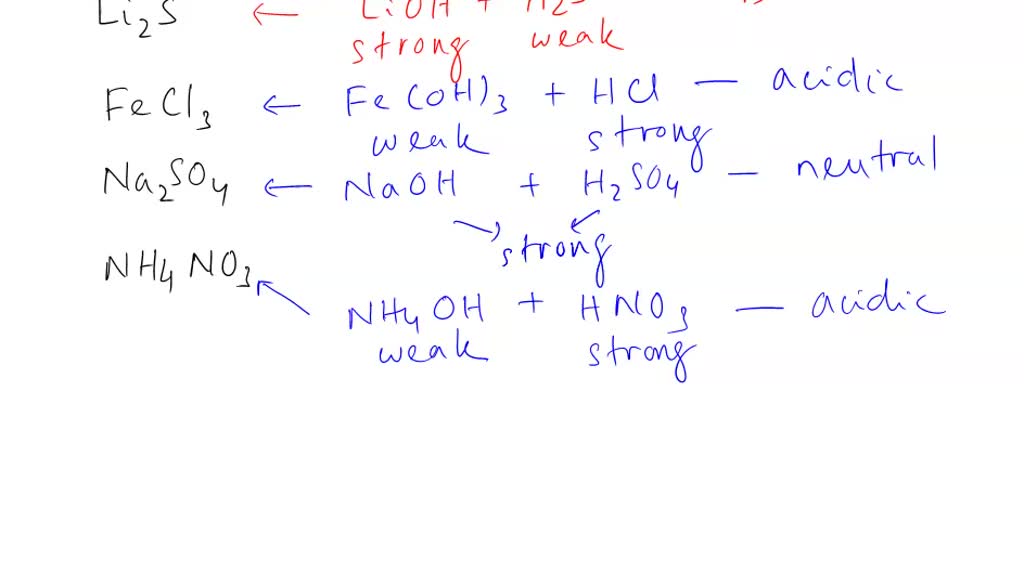
SOLVED: What is the equation for hydrolysis for anion and cation. Determine whether salt is acidic, basic or neutral. 1.) K3PO3 2.) Li2S 3.) FeCl3 4.) Na2SO4 5.) NH4NO3

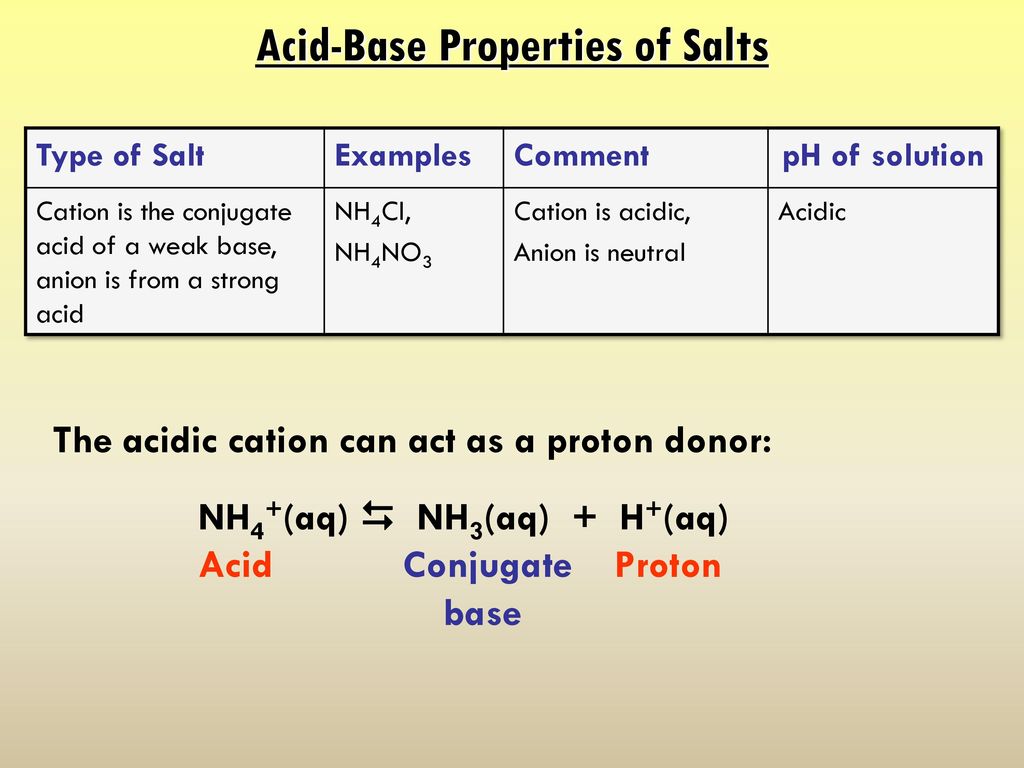
PPT - CHAPTER 10 Reactions in Aqueous Solutions I: Acids, Bases & Salts PowerPoint Presentation - ID:2953050

Ammonium nitrate, NH4NO3, is a salt formed from the neutralization of the weak base ammonia with the strong acid nitric acid. Given that the value of Kb for ammonia is 1.8 x
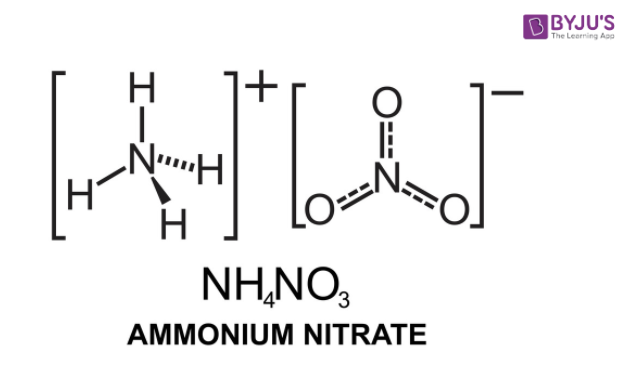
Ammonium Nitrate (NH<sub>4</sub>NO<sub>3</sub>) - Structure, Preparation, Physical and Chemical Properties, Uses with FAQs of Ammonium Nitrate

OneClass: Ammonium nitrate, NH4NO3, is a salt formed from the neutralization of the weak base ammonia...