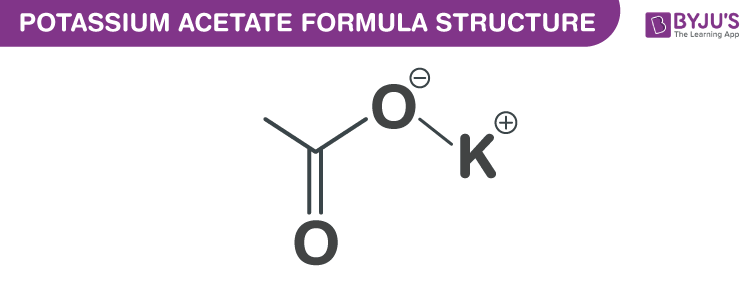
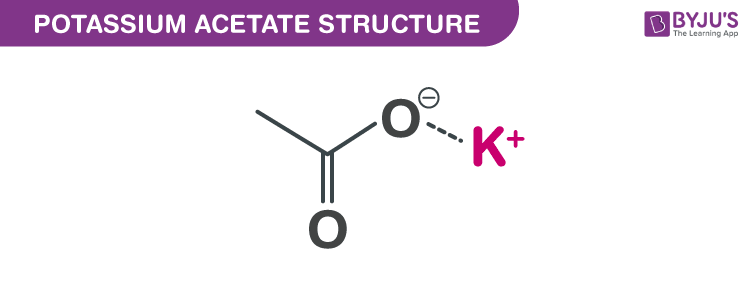
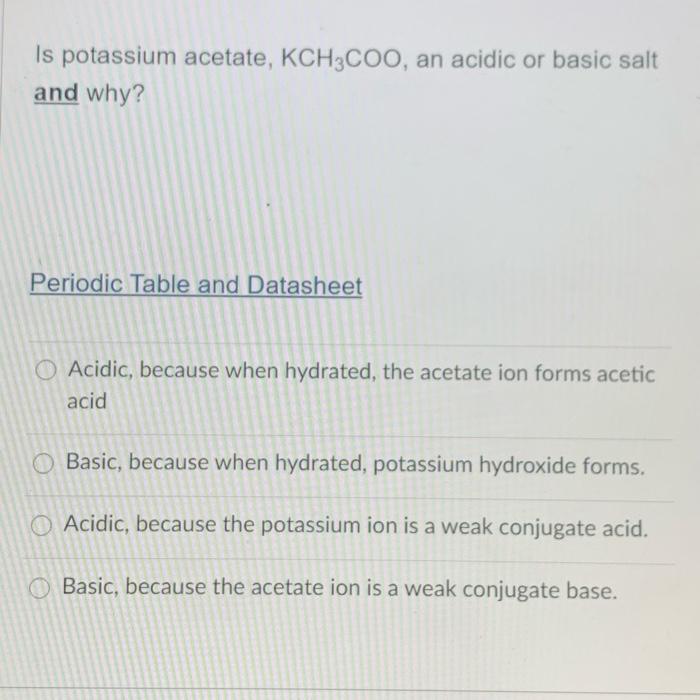
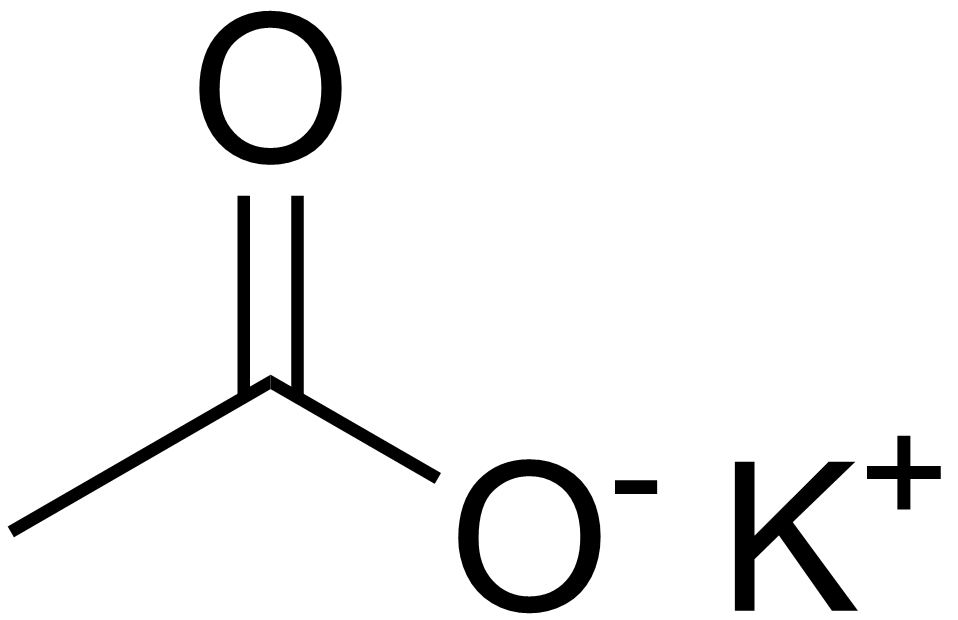
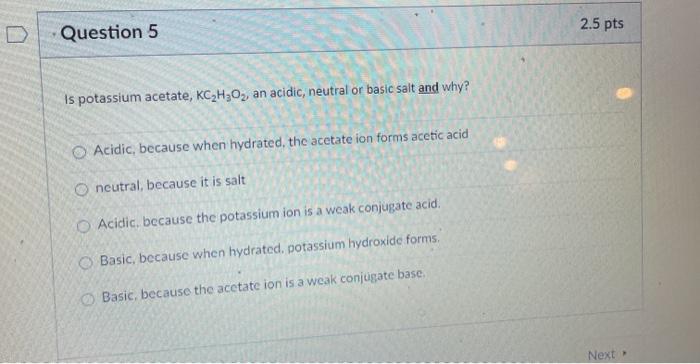
SOLVED: Which of the following explains why a 0.10 M solution of potassium acetate, KC2H3Oz, is less alkaline (basic) than a 0.10 M solution of potassium cyanide; KCN? Cyanides are less soluble

pKa for acetic acid is 4.74 . What should be the ratio of concentration of acetic acid and acetate ions to have a solution with pH 5.74 ?

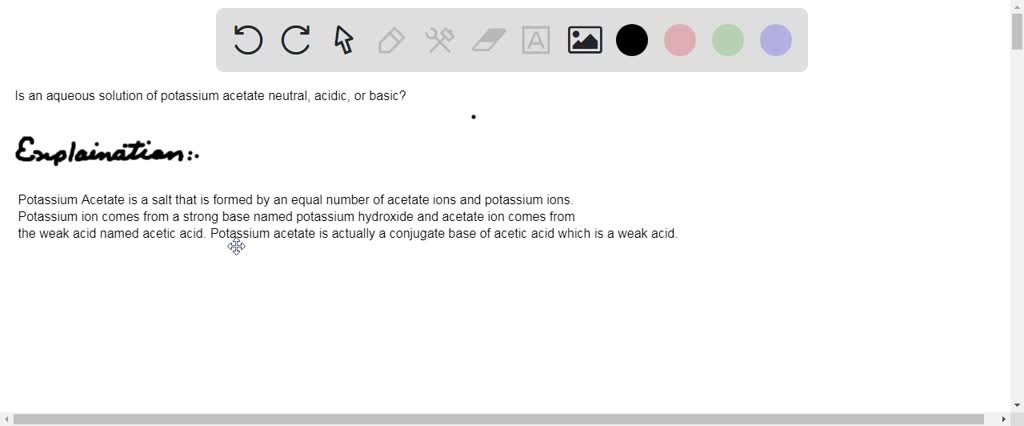
1 Acid-Base Properties of a Salt Solution One of the successes of the Brønsted- Lowry concept of acids and bases was in pointing out that some ions can. - ppt download

Potassium Acetate BP EP USP FCC Acetic Acid Potassium Salt Manufacturers and Suppliers - Price - Fengchen